By Kabuye Ronald
President Yoweri Museveni, on Tuesday afternoon, commissioned Microhaem Scientifics (MHS), a state-of-the-art diagnostic manufacturing facility in Ntinda, near the capital Kampala.

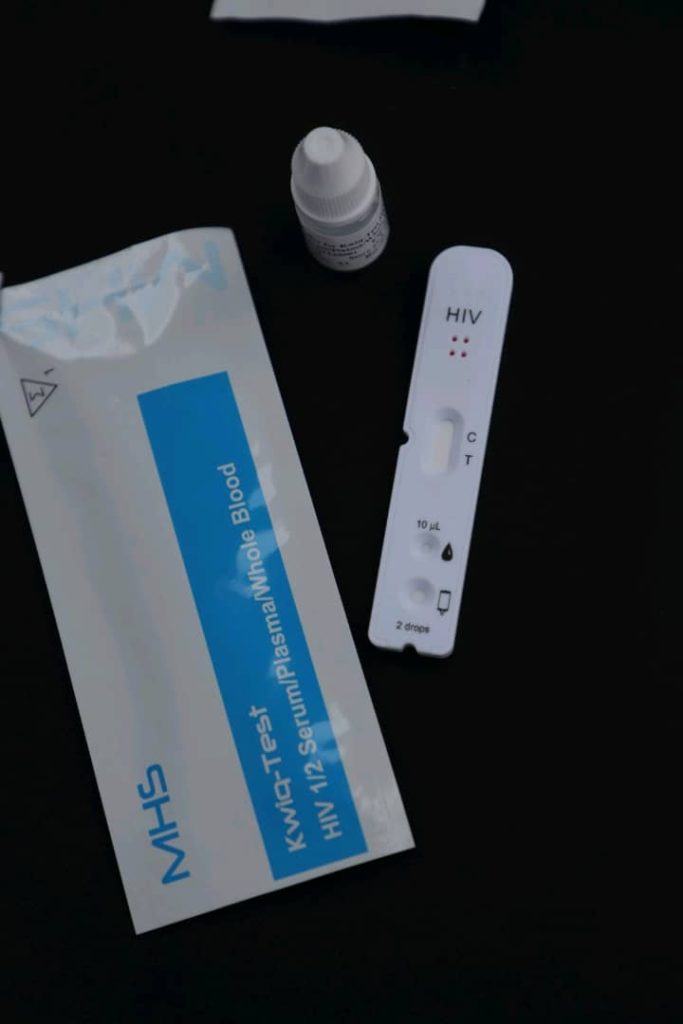
The diagnostic plant, a trailblazer in Sub-Saharan Africa, will produce high-quality yet affordable test kits across a range of diagnostics devices including HIV and malaria test kits that will make a difference to millions of families across Africa.
The president lauded MHS Managing Director Dr. Cedric Akwesigye for establishing the project, adding that it is such investments that will make Uganda a modern economy catalyzed by growth and development.
“I want to salute you young people for waking up and getting out of the slavery of dependency because that has been the problem for Africa,” Mr. Museveni said, urging Ugandan scientists to use their scientific knowledge to solve the many scientific problems affecting the continent.
“This is possible because you really have a very powerful resource, you people. The powerful resource you have is the market. The Market is part of the business, anybody who manufactures, if he cannot sell, then that business is not business,” he said adding that in the next 30 years, Africa’s population will be 2.5 billion.
The facility is also the only Great Lakes Region diagnostic test kits manufacturing plant was guided by National Drug Authority (NDA) and World Health Organisation standards.
The development of the $25 million (about UGX 96 billion) facility has been financed through partner funding from the Uganda Development Bank (UDB), which provided a crucial top-up of USD 6.3 million (about UGX 24 billion) including the importation of Hi-Tech Medical Equipment and machines as well as working capital support.
The president said the government will continue to capitalize UDB, the country’s development bank to enable more Ugandan entrepreneurs to access affordable development capital.
The President assured Dr. Akwesigye that the government through the Ministry of Health will procure the test kits produced by MHS, particularly for HIV and malaria among other essential medical devices, and asked the firm to invest in research to develop more homemade molecular and diagnostic devices.

In a speech at the commissioning of the multimillion-dollar facility, Dr. Akwesigye said that the plant, a toast of Uganda’s science innovation, will serve as the production hub for a diverse range of molecular and diagnostic test kits, encompassing HIV viral load kits, EID kits, Hepatitis B kits, among others.
He revealed that Microhaem Scientifics acquired technology from leading partners including Guangzhou Wondfo Biotech Co., Ltd (China) and Deseret Laboratories Inc (US Based) — marking a significant milestone in the advancement of Uganda’s health sector.
Constructed in strict adherence to WHO standards, including ISO 13485 cGMP, Dr. Akwasigye said this facility has undergone thorough inspection by the NDA and World Health Organisation.
In its initial phase, he announced the production readiness of HIV and Malaria testing kits, meeting the highest international quality benchmarks.

Currently, Uganda’s demand for diagnostics in the public health sector alone is projected at $200 million per year. While funding from development partners and the government combined contributes $117 million, there remains a gap of approximately $83 million per year.
Dr. Akwesigye said the initiative has the potential to bridge the gap and reduce costs.
“To retain this revenue within our economy, we seek government support through off-take guarantees with concrete figures and full commitment, import restrictions (both tariff and non-tariff), and tax exemptions,” Dr. Akwesigye said.
“With this state-of-the-art facility, Uganda can seize this opportunity, significantly boosting our economy. This revenue alone surpasses three times the income generated from coffee exports as of September 2023.”
He asked the president to help facilitate market access in East Africa, particularly Sudan, Congo, Kenya, Tanzania, and Somalia among other African countries.
Dr. Jane Ruth Aceng, Uganda’s Minister of Health congratulated Microhaem Scientifics for leading in the local diagnostic manufacturing space.
“I want to thank the Scientific Advisory Committee for the Microhaem Scientifics group of companies and the international partner investors for accepting to come in and support local manufacturing including technology transfer,” the minister said, telling the president that the Microhaem Scientifics is worthwhile.
“In order for you to appreciate the importance of local manufacturing of medical devices, let me take you a little back to the peak of the COVID-19 pandemic in 2020 and 2021. The Ministry of Health was carrying out a survey to establish the extent of the spread of COVID-19 infection in the population testing and tracking contacts of confirmed COVID-19 cases and testing truck drivers to limit importation of the virus. Suddenly, we’re running out of PCR reagents and supplies, and countries that were manufacturing didn’t want to export them before they could satisfy their own needs. It took the intervention of His Excellency the President talking directly to the heads of the global manufacturing corporations for the country to get some of the laboratory reagents and supplies,” the Minister said noting that “with local manufacturing, we wouldn’t have gone through all these challenges, because we would be having direct access to the reagents and supplies.”
She said that local manufacturing of diagnostics is also in line with a global trend, where the World Health Organization is working with international partners like PEPFAR and the Global Fund to promote manufacturing of the diagnostics locally to ensure sustainability.
“Most importantly for Uganda, local manufacturing is a deliberate plan through our Vision 2040 where we seek to reduce the import bills, have increased trade revenues to accelerate national development and transition to middle-income status.”
Dr. Aceng pledged that the government’s central public health laboratory, which has already been designated by WHO, as a regional center of excellence for diagnostics, will provide quality assurance services to ensure that the products released into the market meet the required quality standards.

